Mycotoxin Effects on Dairy Cattle
by Bill Seglar, DVM, PAS
Nutritional Sciences Manager
Pioneer Hi-Bred Intl., Inc., Box 1150
Johnston, IA 50131-1150
Introduction
Mycotoxins are toxic substances produced by fungi (molds) growing on crops in the field or in storage. Only a few mold species produce mycotoxins out of the thousands of molds that grow on stored grains and forages. While greater than 400 mycotoxins have been chemically identified, the biological or veterinary medical impact of only several mycotoxins is known. (8) The awareness of mycotoxins in forages by dairy producers is greater today than several years ago for several reasons: 1) mycotoxin diagnosis kits are easy and economical to use, 2) enhanced awareness of mycotoxins, 3) modern no-till soil practices, 4) increased dairy production stress, and 5) recent years of inclement weather. (18)
Much confusion results when people discuss mycotoxins in forages. This is because several mycotoxins may exist, diagnostic methods are not consistent, and treatment and control recommendations lack needed research. The mycotoxin identified in forages may not be the causative agent, however an unidentified toxin may be causing livestock problems. Additional confusion results when consultants attempt to discuss the impact of forage mycotoxins on the dairy animal by relying upon the research findings of feeding infected grains to monogastrics.(13) Few mycotoxin investigators have identified the mode of action of mycotoxins within the ruminant. Dairy herds thought to suffer from mycototoxicosis are associated with milk production losses and failure to respond to veterinary therapy and/or changes in nutrition. Symptoms are vague and nonspecific which may include: reduced feed intake, feed refusal, unthriftiness, rough hair-coat, poor body condition, and reproductive problems. Field investigations have associated mycotoxins with increased incidence of fresh cow problems including displaced abomasum, ketosis, retained placenta, metritis, mastitis, and fatty livers.(22)
Field and Storage Molds (8, 21)
The growth parameters of molds vary in that some proliferate while the crop is growing in the field while others propagate during storage.
Field fungi conditions that contribute to their activity include high humidity (>70%) and temperatures that fluctuate between hot days and cool nights. Field molds usually do not grow in stored ensilage because the low pH and oxygen silage environment is not conducive to their survival.
Many plant diseases result from field produced fungi. Examples include ear and stalk rot caused by Fusarium (also known as Gibberella), Diplodia, and Anthracnose, foliar leaf diseases caused by Helminthosporium, and smut caused by Ustilago. (3) Fusarium is associated with mycotoxin production while the other molds do not produce metabolites that are toxic to cattle.
Field mold spores propagate in both grain and forage parts of the plant. Mold spores enter grain through the pollination process via silk channels and invade the endosperm. The physical damage to the kernel pericarp is a second mode of entry, making hard kernel texture a high priority goal for corn breeders (12). Molds may also gain entrance into the forage portion of plants through roots during the
seedling stage from soil contamination, or from physical damage to plant tissue from hail and insects. (11) Bt hybrids should lower the amount of mold/mycotoxin activity in corn hybrids and is an area of present research. However, research findings are inconsistent in that Mulkvold from Iowa State shows Bt hybrids to have lower levels while Pioneer research shows no difference between Bt and parent genetics.
Storage fungi usually do not invade the crop prior to harvest. These soil-borne mold spores are brought into the silo with forages. Up to 24 molds have been identified in ensilage, however most are considered non-producers of mycotoxins (10, 23). The most prevalent molds isolated from most North American silages are Mucor, Penicillium, Aspergillus, and Monillia. Aspergillus flavus may produce aflatoxin as a field mold, although it is classified as a storage organism.
Some silage molds can grow within low oxygen and moderately low pH environments; however, their survivability is limited to competition with anaerobic bacteria. Therefore, more stable silages are less prone to become moldy (13). Usually mold activity is initiated from an elevated pH due to aerobic lactic acid consuming yeast that become active from the introduction of oxygen into the silo. Candida and Hansula are lactate consumers; these organisms usually preclude mold activity when their population counts exceed 100,000 colony-forming units per gram of forage (CFU/Gm) (10).
Mycotoxins of Concern (8)
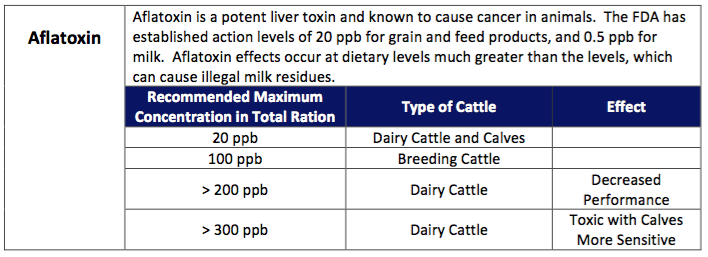
The commonly diagnosed mycotoxins are aflatoxin, which is produced by Aspergillus flavus, DON (vomitoxin), zearalonone, T-2, and fumonisin, which is produced by Fusarium sp. Aflatoxin is commonly found in the southern United States and is a major public health concern because it’s carcinogenic properties to humans. It’s the only toxin in which the FDA has imposed limitations regarding how much may be fed in dairy rations. The current surveillance programs aimed at reducing food residues make it very unlikely for aflatoxin to have significant production or health effects on dairy herds. Usually identification of aflatoxin from northern grown forages is due to “false-positive” results associated with the ELISA method of laboratory detection.
Deoxynivalenol (DON) is the proper name for the most often detected Fusarium mycotoxin, often referred to as vomitoxin. Specific modes of action have been identified in swine that explains this toxin as the primary cause of feed refusals, diarrhea, vomiting, reproductive failures, and death. However, DON in cattle has only been associated with reduced feed intake and lower milk production. Whitlow from North Carolina collected clinical data from 300 herd representing about 40,000 cow records showing that DON was associated with a loss in milk production, but did not establish a cause and effect mode of action. DON may simply be a marker for the presence of other mycotoxins in problem feeds.

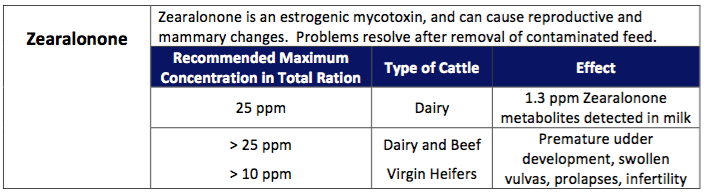
Zearalonone is a Fusarium produced mycotoxin that elicits an estrogenic response in monogastrics. Usually it’s found secondary to and at much lower levels than DON. Ruminants can ruminally degrade zearalonone and therefore is less toxic to dairy cattle. Controlled studies have added up to 22 ppm (parts per million) to dairy rations and failed to generate reproductive problems. Field observations of poor intakes, depressed milk production, and reproductive problems have been associated with the presence of DON and zearalonone. This may be attributed to the presence of other mycotoxins or interaction with other factors.
T-2 toxin is another Fusarium produced mycotoxin and is seldom detected in forages of the upper Midwest. T-2 studies document that it causes gastroenteritis in laboratory animals. Cattle studies do exist that associate the toxin with feed refusal and diarrhea along with immunosuppression in dairy calves.

Fumonison is another Fusarium produced mycotoxin, however it’s more commonly found in the southern United States, compared to DON, zearalonone, and T-2 which are prevalent in the northern states. Fumonisin is a proven cancer promotor and modes of action have been identified to explain its effects on horses and monogastrics. The toxin causes liver damage and decreased milk production in dairy cattle at levels greater than 100 ppm. Usually levels in feeds exist at the 1-10 ppm range.

Other mycotoxins exist that are not included in diagnostic screenings (19), which include CPA produced from Penicillium and Aspergillus spp., PR toxin from Penicillium rouquefortii, patulin from Penicillium spp., AAL from Alternaria, and fusaric acid from Fusarium spp.
Mode of Actions Associated with Mycotoxicosis
An epidemiology survey (19) conducted in 1997 showed that a high or low test for DON in silage in many cases is not conclusive in explaining the absence or presence of problems in almost half of reported cases throughout Wisconsin, Vermont, and New York. This leads researchers to postulate that another toxin (or toxins) is present in the silage. This toxin is either working synergistically with DON, or, DON is often present coincidentally because several different contaminating fungi may have similar environmental requirements for production of different toxins. This is true for the fungi Fusarium and Penicillium that produce DON and roquefortine toxins respectively. Farms may have healthy animals can also have high levels of DON when tested.
Ruminants appear relatively tolerant of the adverse effects of mycotoxins and perhaps are due to the ability of the rumen microflora to detoxify these toxins. However increased rumen passage rates of today’s high production dairy cattle likely overwhelm the ability of the rumen to completely denature the toxins. Production and health problems from cattle consuming mycotoxin-infested feeds probably are enhanced by a mixture of toxins resulting in a synergistic interaction (13). May, Wu, and Blake (24) at South Carolina looked at the effect of DON and fusaric acid in ruminal in vitro digestion experiments. They found that when DON was present by itself, there was no effect on microbial protein synthesis. However, DON and fusaric acid present together caused depressed Ruminococcus albus and Methanobrevibacter ruminantium microbial activity.
Researchers from University of Minnesota (20) recently looked at the effects of Penicillium produced patulin, because of prior findings and because it has been related to diseases in cattle fed on moldy fermented feeds in England, Japan, France, and Germany. The objective of this study was to determine the dose-response effect of patulin exposure on fermentation by ruminal microbes maintained in continuous culture fermenters.
Eight single-flow continuous in vitro culture fermenters were inoculated with ruminal fluid from a cannulated cow. Substrate for microbial metabolism was provided by incorporating 75 g of DM/d of a pelleted diet via an automated feeding device. The diet contained 38% alfalfa hay, 28% corn silage, 27% cracked corn, 5% soybean meal, and 0.6% of a mineral mix (DM basis). Chemical composition of the diet (% of DM) was 92.9% OM, 15.5% CP, 27.4% NDF, 16.2% ADF. The experiment was conducted during two 7-day experimental periods. After four days of adaptation, two fermenters each were spiked with 0, 30, 60 and 90 ppm of patulin in one ml of distilled H2O every 12 hours for 3 consecutive days. The addition of patulin to continuous culture fermenter flasks had the following effects on microbial metabolism:
- Digestion of true organic matter, acid detergent fiber and crude protein was reduced (P < .05) by approximately 27, 43 and 36%, respectively with patulin addition ranging from 30 to 90 ppm.
- Total volatile fatty concentration (mM) in fermenter effluent decreased (P < .05) from 180.1 to 119.7 with the addition of 90 ppm of patulin but did not differ (P > .05) between the control treatment and 30 to 60 ppm addition of patulin.
- Acetate (mol/100 mol) was depressed (P < .05) with patulin addition due to a reduction in fiber digestion. Conversely, there was a shift in fermentation with butyrate and valerate (mol/100 mol) increasing (P < .05) with the addition of patulin.
- At the highest levels of patulin addition (60 and 90 ppm), branched-chain VFA were lower (P < .05) resulting from a reduction in protein (branched-chain amino acid) degradation.
- Bacterial nitrogen flow was lower (P < .05) with patulin addition (30 to 90 ppm) compared with the control treatment, while the efficiency of bacterial growth (g of N/kg OMTD) was lowest (P < .05) when 90 ppm of patulin were added to fermenter flasks.
From this experiment, it was concluded that patulin can alter metabolism of nutrients by ruminal microbes. Adverse effects of patulin include a reduction in organic matter, fiber and protein digestion resulting in changes in fermentation end-products and nutrient flow. These changes can potentially have a negative impact on animal health and performance
Another experiment looking at the effects of Penicillium produced toxins is being led Dr. Alan Gotlieb from the University of Vermont, which began last year. 200 silage samples were collected from 80 dairy herds in which Penicillium isolates of four different species were consistently identified as being able to tolerate an acid pH environment in the 4.0 range. A commercial Thin Layered Chromatography technique was used to identify 8 mycotoxins, of which roquefortine was the most common isolated toxin. Laboratory techniques have been successfully developed to grow the Penicillium isolates on sterile corn media in a pH environment less than 4.0. The next step in this investigation will be to subject roquefortine infected silages to in vitro tests to determine if microbial activity is diminished by the presence of the toxin in corn silage.
Treatment and Management of Contaminated Silages (18)
Remediation techniques exist for denaturing mycotoxin infested grains by using direct flaming, anhydrous ammonia, or sodium bicarbonate methodologies. However, none of these techniques work on mycotoxin contaminated forages (15).
While mycotoxins cannot be denatured in silages, the forages can be managed to minimize livestock problems. One practical way to minimize the effects of toxins is to dilute affected feedstuffs with clean feed.
Adsorbants are used to “bind” mycotoxins found in feeds. Many of these products have FDA approval for aflatoxin, however are not approved for other toxins such as DON. Some manufacturers have limited research suggesting binding capability of DON. However, adsorbants used for DON are generally marketed as feed additives having anti-caking properties or direct-fed microbial benefits.
Other “shotgun” approaches that have been used by consulting nutritionists to help neutralize the effect of toxins include increasing the ration level of energy, protein, vitamins (A, E, B-1) and minerals (Se, Zn, Cu, Mn).
Continual monitoring of mycotoxin levels is advisable when infected forages are fed. Mycotoxin levels may drastically change depending on where harvested forages were harvested within the environment. Veterinary and nutritional professionals should be persistent in the formation of a complete differential diagnosis regarding the cause of herd health and production problems. Often mycotoxin is blamed as the primary cause of dairy problems, when other factors (or combination of factors) were involved.
Control of Silage Molds/Mycotoxins (18)
Most “commonly diagnosed” mycotoxins found in forages are produced in the field before ensiling occurs. However, evidence indicates that storage molds may produce “uncommonly diagnosed” toxins in the storage structure. Therefore, prevention of mold and mycotoxins involves working with the crop while growing the field and during storage in the silo. Employing all control measures isn’t always possible, however the application of some of the ideas may minimize the level mold and mycotoxin activity.
Plant pathologists indicate that planting early and harvesting early seems to result in lower mold activity in the crop and lowers chances of mycotoxin production. Mold spore levels may be higher with no-till soil management. Moldboard plowing or deep disking is advised to facilitate the degradation of crop debris that can fuel mold growth.
Optimizing soil fertility to improve plant health can reduce mold activity. Producing healthy plants helps diminish plant stresses such as stalk lodging and corn borer induced channels for molds to enter the ear. Anything that helps improve plant health will likely help reduce disease lesions and pest damage, thus suppressing mold invasion and mycotoxin production.
Some researchers theorize that planting corn year after year on the same ground creates the opportunity for increasing mold levels. Most plant pathologists advise crop rotation (with Fusarium resistant crops) to “break the cycle” on susceptible farms.
Corn silage growers should consult their seed supplier for disease resistance ratings on specific hybrids that includes: 1) Gibberella ear rot, 2) Fusarium ear rot, and 3) Anthracnose stalk rot. Corn breeders and pathologists employed by leading seed genetics companies are scoring inbreds and hybrids for mycotoxin resistance. However, limited databases prohibit the current marketing of hybrids based upon mycotoxin resistance claims. Future seed products will likely have separate fumonisin and DON resistance claims for select hybrids.
Hybrid selection should include a range of harvest maturities to spread out the risk of mold/mycotoxin activity during the silking period. In addition, hard kernel textured hybrids are essential to minimize mold/mycotoxin production while in the field. Crop stressors such as bird, hail, flood, or insect damage will increase the chances for mold growth.
A timely harvest at proper moisture and maturity levels not only ensures that Fusarium activity will be minimized in the field, but also that storage mold activity will be minimized within the silo. Other silo management considerations that promote optimal fermentation will also minimize mold/mycotoxin during storage. These include: 1) fast fill, 2) proper chop length, 3) proper packing, and 4) covering the silo to reduce exposure to oxygen. Additionally, making sure the silo is clean prior to filling and that mud and manure is eliminated during the ensiling process will minimize the mold spore load entering during filling of the silo.
The incorporation of well-researched bacterial inoculants will ensure optimal fermentation to quickly reach terminal pH and ensure stable silage. The use of inoculants, or any other silage additive, will not detoxify mycotoxins.
Finally, proper feedout of the silo is a must to minimize aerobic instability and the initiation of mold growth. The analyses of moldy silages usually reveal that the preserved silage mass was stable. However, moldy silage usually is results when producers fail to maintain proper feedout rates, utilize proper feedout methods, and keep the face clean.
Conclusions
There are several key points to remember when dealing with potential mycotoxin problems in animal feeds:
- Most molds are harmless in that they do not produce known mycotoxins.
- The majority of commonly diagnosed mycotoxins are produced in the field prior to harvest.
- Vomitoxin (DON) should be considered a “marker”; if it is present, conditions exist for the growth of their potential toxin producers.
- ELISA tests should be backed up by HPLC or GC tests since current ELISA tests given many false positives when used in silage.
- If a problem is suspected, a comprehensive differential diagnosis is essential – e.g.: many herd problems blamed on mycotoxins turn out to be nutritional.
- Proper crop management, from field to feedout, can reduce the opportunities for mold growth and subsequent toxin production.
References
- Adams, R.S., et al, 1993. Mold and Mycotoxin Problems in Livestock Feeding. Penn State, College of Ag. Sc. Coop Ext., DAS 93-21.
- Bellman, Et al, 1992. Silo Operator’s Manual. Intl Silo Assn. Inc., p. 5-10
- Bundy, L., Carter, P., Doersch, R., Grau, C., Wedberg, J., 1993. Pollination to Maturity. Corn Management/ Diagnostic Guide, Pioneer Hybrid Intl., Inc. pp. 33-34.
- Casteel, S.W., Rottinghaus, G.E. 1995. Liver Disease in Cattle Induced by Consumption of Moldy Hay. Vet. Human Toxicol. 37:248-250.
- Charney, E., Trenholm, H.L., Thompson, V.D., Nicholson, D.B.., Prelusky, L.L. 1993. Influence of Level of Deoxynivalenol In the Diet of Dairy Cows on Feed Intake Milk Production, and its Composition. J. Dairy Sc. 76:3580.
- DiCostanzo, A., Johnston, L.F., Murphy, M., 1995. Effects of Molds on Nutrient Content of Feeds Reviewed. Feedstuffs, Jan 16, 1995, p. 17-54.
- Dodd, J.L., 1983. Corn Stalk Rot: Accounting for Annual Changes. 38th Annual Corn and Sorghum Research Conference. Am. Seed Trade Association. P. 71-74
- Kore, A.N., 1996. Mycotoxins in Animal Feeds: Current Issues and Concerns. WI Veterinary Medical Assn. Proceedings 81:207-214.
- Mahanna, W.C., 1993. Troubleshooting Silage Problems. 4-State Applied Nutrition Conf. Proceedings, LaCrosse, WI. Table 9.
- McDonald, P., Henderson, A.R., Heron, S.J. E., 1991. The Biochemistry of Silage, 2nd Ed. Clalcombe Publications, 13 Highwoods Drive, Marlow Bottom, Marlow, Bucks SJ&3PU pp. 129-130.
- Miller, J.D., Young, J.C., Trenholm, H.L., 1983. Fusarium Toxins in Field Corn. I. Time Course Of Fungal Growth And Production Of Deoxynivalenol And Other Mycotoxins, Can. J. Bot. 61:3080- 3087.
- Miller, J.D., 1994. Fungi in Grain: Implications for Stored Product Research. J. of Stored Products Research, Vol. 31, No. 1, pp. 1-16.
- Miller, J.D., Trenholm, H.L., 1994. Mycotoxins in Grain: Compounds Other Than Aflatoxin, 1st Ed. Eagan Press, 3340 Pilot Knob Rd., St. Paul, MN pp. 302-303.
- Ohmomo, S., Kitamoto, H.K., 1994. Detection of Roquefortines in Penicillium roquefortii Isolated from Molded Maize Silage. J. Sci. Food Agric., 64:211-215.
- Park, D.L., 1993. Perspectives on Mycotoxin Decontamination Procedures. Food Additives and Contaminants 10:49-60.
- Romer, T., 1995.1995 Resource Guide. Romer Labs., 1301 Stylemaster Dr., Union MO.
- Ru-Dong, W., Still, P.E., Smalley, E.B., Schnoes, H.K., Strong, F.R., 1972. Isolation and Partial Characterization of a Mycotoxin from Penicillium rouquefortii. Applied Microbiology, Jan,199\73, p. 111-114.
- Seglar, W.J., Mahanna, W.C., 1995. Mold and Mycotoxin Update. Pioneer Hi-Bred Intl, Inc. Box 1100, Johnston, IA 50131.
- Seglar, W.J., Blake, C., Chu F.S., Gotlieb, A.R., Thomas, C., Thomas, E., Trenholm, H.L., Undersander, D.,1997. Comparison of Mycotoxins Levels Among Problem and Healthy Dairy Herds. Unpublished. Pioneer Hi-Bred Intl., Inc. Des Moines, IA
- Tapia, M.O., Murphy, M.J., Koski, R.L., Stern, M.D. 2000. Influence of Patulin Level on Fermentation by Ruminal Microbes in Continuous Culture. University of MN, College of Veterinary Medicine, St. Paul, MN.
- Watson, S.A., Ramstad, P.E., 1994. Corn: Chemistry and Technology, 3rd Ed. AACC, 3340 Pilot Knob Road, St. Paul, MN. P. 159, 439.
- Whitlow, L.W., Hagler, W.M., 1998. The Potential for an Association for Mycotoxins with Problem of Production, Health, and Reproduction in Dairy Cattle. Proceedings MN Dairy Health Conference, May 19-21, 1998. College of Vet Med., UM, St. Paul, MN.
- Woolford, M.K., 1984. The Silage Fermentation. Marcel Dekker, Inc. 270 Madison Ave., Ny York, NY 10016 P32-33.
- May H.D., Wu Q, Blake C.K. 2000. Effects of the Fusarium spp. Mycotoxins Fusaric Acid and deoxynivalenol On The Growth of Ruminococcus albus and Methanobrevibacter ruminantium. Canadian J. of Microbiology. V. 46. P.692-699. 2000.